 Summary
Summary
Contributing author
John R Evans
1. Summary – A-Ci Curves
Definitions
A-Ci Curves The underlying biochemistry of a leaf can be probed by measuring the response of CO2 assimilation rate to intercellular CO2 partial pressure. The latter is varied by changing the CO2 partial pressure in the air surrounding the leaf during measurement. The intercellular CO2 response curve allows one to separate the influence of stomata from underlying biochemical limitations.
Terminology and equations
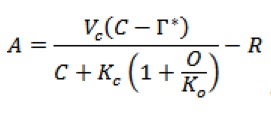 (1)
(1)
 (2) A rate of CO2 assimilation (μmol CO2 m-2 s-1) Vc is the maximum Rubisco carboxylase activity (μmol CO2 m-2 s-1) (its activation state can vary with light, CO2 and temperature) Kc and Ko are the Rubisco Michaelis Menten constants for CO2 and O2 O is the partial pressure of oxygen (mbar) J is the rate of electron transport (μmol e– m-2 s-1) C is the partial pressure of CO2 at the sites of carboxylation within the chloroplast (Cc), but intercellular CO2 partial pressure (Ci) can be used. Cc = Ci – A/gm where gm is the mesophyll conductance to CO2 between the intercellular airspaces and the sites of carboxylation within the chloroplast. * CO2 photocompensation point (μbar) R day respiration rate (μmol CO2 m-2 s-1)
(2) A rate of CO2 assimilation (μmol CO2 m-2 s-1) Vc is the maximum Rubisco carboxylase activity (μmol CO2 m-2 s-1) (its activation state can vary with light, CO2 and temperature) Kc and Ko are the Rubisco Michaelis Menten constants for CO2 and O2 O is the partial pressure of oxygen (mbar) J is the rate of electron transport (μmol e– m-2 s-1) C is the partial pressure of CO2 at the sites of carboxylation within the chloroplast (Cc), but intercellular CO2 partial pressure (Ci) can be used. Cc = Ci – A/gm where gm is the mesophyll conductance to CO2 between the intercellular airspaces and the sites of carboxylation within the chloroplast. * CO2 photocompensation point (μbar) R day respiration rate (μmol CO2 m-2 s-1)
Measurement approaches
CO2 response curves are measured by varying the partial pressure of CO2 surrounding the leaf and calculating C from stomatal conductance measurements. If mesophyll conductance (gm) can be measured (see Pons et al. 2009), then Cc, the partial pressure at the site of carboxylation can be calculated. The response curve is best measured by either increasing or decreasing CO2 as this prevents -kinks’ forming at the point where curves obtained by combining measurements from two different directions meet. The data can be fitted to the Farquhar, von Caemmerer and Berry (1980) model of C3 photosynthesis to derive values for maximum Rubisco activity (Vc) and the rate of electron transport (J). J and Vc tend to covary.
Ranges of values
The values for Rubisco kinetic parameters are temperature dependent. Comprehensive datasets are limited to only a few species, such as tobacco. See Sharkey et al. 2007 for an excel spreadsheet to fit the model to data.
Related pages
To find out more about curve fitting and the statistical analysis of A-Ci curves, visit the page on A-Ci curves statistics.
2. Summary – Light response of photosynthesis
Definition
Photosynthesis is the process which captures the energy in light and converts it into chemical energy. The rate of photosynthesis varies as the amount of light absorbed varies in a curvilinear response.
Terminology and equations
Irradiance: Flux of photons per unit area Photosynthetic photon flux density PPFD: Flux of PAR photons per unit area Photosynthetically active radiation PAR: photons in the 400-700nm waveband Quantum yield or photon yield: The ratio of photosynthetic rate to irradiance (incident or absorbed, generally PAR) ;:
 (1) ;:
(1) ;:
 (2) J is the rate of electron transport (μmol e– m-2 s-1) maximum photon yield (mol e– (mol absorbed photon)-1) (this varies with wavelength) I irradiance (μmol PAR photons m-2 s-1) curvature factor Jm maximum rate of electron transport per unit leaf area (μmol e– m-2 s-1) A rate of CO2 assimilation per unit leaf area (μmol CO2 m-2 s-1) C CO2 partial pressure at the sites of carboxylation within chloroplasts (this can also be expressed as a concentration, or could be intercellular CO2 partial pressure) * CO2 photocompensation point R day respiration rate Equation 1 describes a non rectangular hyperbolic response of electron transport rate to irradiance. Equation 2 converts J to A, by accounting for photorespiration.
(2) J is the rate of electron transport (μmol e– m-2 s-1) maximum photon yield (mol e– (mol absorbed photon)-1) (this varies with wavelength) I irradiance (μmol PAR photons m-2 s-1) curvature factor Jm maximum rate of electron transport per unit leaf area (μmol e– m-2 s-1) A rate of CO2 assimilation per unit leaf area (μmol CO2 m-2 s-1) C CO2 partial pressure at the sites of carboxylation within chloroplasts (this can also be expressed as a concentration, or could be intercellular CO2 partial pressure) * CO2 photocompensation point R day respiration rate Equation 1 describes a non rectangular hyperbolic response of electron transport rate to irradiance. Equation 2 converts J to A, by accounting for photorespiration.
Measurement approaches
Light drives the rate of electron transport and subsequently the rate of RuBP regeneration which is consumed by Rubisco when catalyzing carboxylation or oxygenation reactions. Equation 1 describes the curvilinear response of electron transport in the absence of any other limitation. Chlorophyll fluorescence provides an optical method to measure photochemical efficiency which can be used to calculate J. Photosynthesis can also be measured using an oxygen electrode, generally under saturating CO2 conditions, where the oxygen evolution rate equals J/4. At lower CO2 partial pressures, the activity of Rubisco may impose a lower maximal rate and truncate the response curve. Photosynthesis is frequently measured as CO2 assimilation, so equation 2 converts the electron transport rate into a CO2 assimilation rate by accounting for photorespiration (the oxygenase reactions of Rubisco) and other mitochondrial respiration. Many photosynthetic enzymes are light regulated, so following a dark period, photosynthesis requires an induction period before reaching steady state. During this time, stomata may also be opening. Consequently, light response curves may vary depending on whether the measurement sequence is made with increasing or decreasing irradiance. The dependence on CO2 partial pressure means that attention must be paid to ambient CO2 partial pressure and stomatal conductance. Mitochondrial respiration rates continue in the light, generally at a lower rate than that in the dark. Extended periods under high irradiance can lead to photoinhibition which decreases the rates at lower irradiance and is characterized by a reduction in . This may recover within minutes or may require days to repair, depending on the mechanism. When fitting Eq. 1, one should be aware that interacts with . It may be necessary to fix to a reasonable value. The absorptance of a leaf is the fraction of incident light that is not reflected or transmitted. For PAR, leaf absorptance generally ranges between 0.7 and 0.95 and can be measured with an integrating sphere.
Ranges of values
0.425-0.5 mol e– (mol absorbed PAR photon)-1 0.7-.96 Jm 50-200 μmol e– m-2 s-1 R 0.1-3 μmol CO2 m-2 s-1 I 0-2300 μmol PAR photons m-2 s-1
Related pages
An Excel spreadsheet to help speed up A/Ci calculations is available here
Health, safety and hazardous waste disposal considerations
There are no health and safety considerations associated with this summary, however it is recommended that you consult the relevant MSDS before using any chemicals in related experiments.
Literature references
Evans J.R., Jakobsen I. & Ögren E. (1993) Photosynthetic light response curves. 2. Gradients of light absorption and photosynthetic capacity. Planta, 189, 191-200.
Farquhar G.D., von Caemmerer S. & Berry J.A. (1980) A biochemical model of photosynthetic 2 assimilation in leaves of C3 species. Planta, 149, 78-90
Ögren E. & Evans J.R. (1993) Photosynthetic light response curves. 1. The influence of CO2 partial pressure and leaf inversion. Planta, 189, 182-190.
Pons T.L., Flexas J., von Caemmerer S., Evans J.R., Genty B., Ribas-Carbo M. & Brugnoli E. (2009) Estimating mesophyll conductance to CO2: methodology, potential errors, and recommendations. Journal of Experimental Botany, 60, 2217-2234.
Sharkey T.D., Bernacchi C.J., Farquhar G.D. & Singsaas E.L. (2007) Fitting photosynthetic carbon dioxide response curves for C-3 leaves. Plant Cell and Environment, 30, 1035-1040.
von Caemmerer S. & Farquhar G.D. (1981) Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta, 153, 376-387.
von Caemmerer S. (2000) Biochemical models of leaf photosynthesis (vol. 2). CSIRO publishing, Collingwood, Victoria (Australia).