 Protocol
Protocol
Author
Agata Rascio
Additional AUTHORS
Eleonora Carlino, Giuseppe Nicastro, Cristiano Platani
OVERVIEW
This protocol outlines a rapid method to estimate the tissue affinity for strongly bound water (Rascio et al., 1997), by measuring the differences in water sorption (DWS) by the leaves at two temperatures. Large DWS values are indicative of a high tissue strength of water binding.
BACKGROUND
Analysis of moisture sorption isotherms provides quantitative and qualitative determination of water status that is related to biological differences in the substrate. The construction of isotherms takes time and is labour-intensive. The results are also affected by the equipment design and the handling procedures. Starting from the standardisation of isotherm measurements guidelines of COST 90 (Wolf et al. 1985), which are the most accepted and standardised methods for the measurement of the water activity of a sample, this protocol allows an estimation of the differential enthalpy of strongly bound water, calculated from the sorption isotherm curves from van’t Hoff analysis. The low cost of the salt (magnesium chloride) used for the static control of the relative humidity, the drastic reduction in labour, and the possibility of distributing analyses of stored dry samples over a convenient period of time, make this DWS analysis a very useful method to test a large number of samples.
MATERIALS/EQUIPMENT
- Desiccator (30-cm diameter), with silicon grease and vacuum pump;
- Sorption apparatus: glass jars (1 l) with tripod and wire mesh, closed by a rubber sealing ring;
- Phosphorus pentoxide P2O5– a desiccant
- About 500 mg freshly collected leaf samples
- Saturated salt solution of Magnesium chloride (MgCl2)
- Analytical Balance (0.0001 g )
- Thermostatic room or water bath (∓1∘C or less)
- Chromic acid-sulphuric acid mixture (10%)
- Pyrex weighing bottles, 20 mm x 20 mm (5 g weight).
UNITS, TERMS, DEFINITIONS
Strongly bound water
Differences for Water Sorption (DWS) ( mg H2O g-1)
Differential enthalpy ( ) of water sorption (kjoules mol -1)
Moisture sorption isotherms
EW= equilibrium net weigh of samples equilibrated with saturated salt solution (g)
DW = equilibrium net weight of samples equilibrated with P2O5 (g)
PROCEDURE
Operating the DWS determination
- Clean two weighting bottles (10g weight) for each leaf sample, by immerging them in 10% chromic acid – sulphuric acid mixture and wash accurately with deionised water.
- Dry the weighing bottles overnight at 105 ∘C. Put the numbered weighing bottles, with the lids open, in a dryer containing P2O5for 1h. Remove them, and weigh each of them rapidly for the empty calibration. Store the weighing bottles in the dryer at room temperature.
- Prepare 500 ml of a saturated solution of MgCl2, adding the salt to boiling distilled water, until no more salt dissolves in the solution (i.e. saturated solution). Maintain a layer of solid salt at the bottom of the saturated solution during the whole period of the equilibration, to be sure that the solution always remains saturated. There should be 4 mm of solution over the crystal layer when it has cooled.
- Set up two sorption apparatus systems. Each of these consists of a 1-litre-capacity glass jar (11-cm diameter), hermetically sealable with a rubber seal and a locking clamp. Insert a tripod into the jar and put a wire mesh on it. Add the saturated salt solution of MgCl2, two cm deep, so that the tripod remains half submerged.

- Equilibrate a closed apparatus, to the constant temperatures of 5∘C and the other one at 20∘C, by storing it in a thermostatic cabinet, room, or water bath. The temperature should be regulated at least six days before the beginning of the leaf incubation.
- Weight about 500 mg samples of fresh leaves.
- Cut the leaves transversally with a razor blade, to obtain large strips of 3.2 cm2.
- Prepare two sub-samples, and insert them separately into the calibrated weighing bottles described in point 2, and leave the bottles open in the dryer containing the phosphorus pentoxide (P2O5), under vacuum. After 96 h, remove and record the mass of the closed bottles.
- Transfer the open weighing bottles into the sorption apparatus described in point 4. Seal the jars. Be sure that the seal is tight enough to prevent leakage of gas from the jar.
- Equilibrate the first sorption apparatus at 5 ∘C, and the second at 20 ∘C in the thermostatic environment. During the incubation, both temperatures must be regulated precisely (∓1∘C) at their constant temperatures. After 96 h, remove and record the mass of the closed bottles.
Calculate DWS, expressed as mg H2O g-1dry matter:
DWS = {(EW5°-DW5°)/DW5° – (EW20°-DW20°)/DW20°}*1000
where:
EW5° = equilibrium net weigh of sample, incubated at 5 ∘C,
DW5° = equilibrium net weight of the same sample after incubation with P2O5
EW20° = equilibrium net weigh of sample, incubated at 20 ∘C,
DW20° = equilibrium net weight of the same sample after incubation with P2O5
Download Excel spreadsheet to calculate DWS here
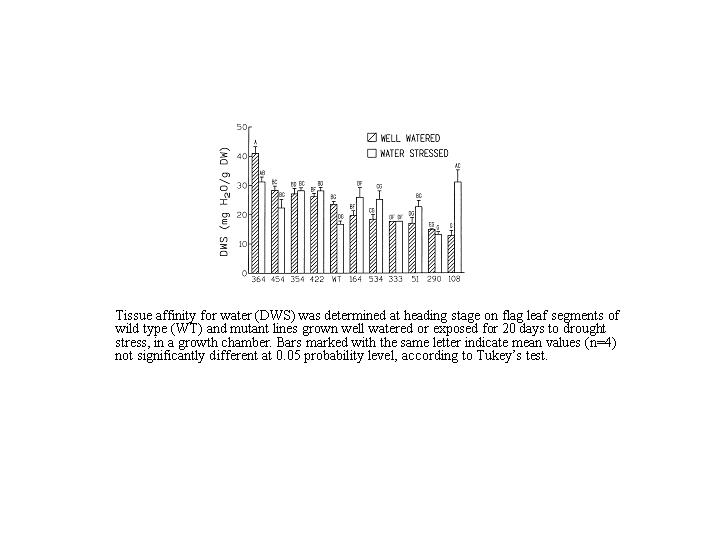
Example data for DWS values for plants obtained from mutagenised wheat seeds.
NOTES AND TROUBLESHOOTING TIPS
The chromic acid-sulphuric acid mixture should be prepared inside a chemical fume hood, because it is toxic and carcinogenic. To clean the weighing bottles, you can substitute this cleaning solution with an ultrasonic bath or biodegradable surfactants.
Four days is the equilibrium time for durum wheat leaves. For different materials, establish that equilibrium has been reached by weighing the samples at intervals, until there is no change in mass.
The simultaneous equilibration of samples at different temperatures requires separate cabinets, although if they are not available, from point 9 onwards the procedure can be delayed. The samples to be incubated at the higher temperature can be equilibrated with the salt solution later, by extending their time with the P2O5.
To obtain replicates, three weighing bottles can be placed in the same jar and/or several jars can be placed in the same cabinet or water bath; alternatively, the experiments can be performed by repeating the above procedure 3-8 times.
DWS allows detecting differences for water binding mode, among different samples, for comparative purpose. For wheat leaves, estimate the average differential hydration enthalpy ( H)expressed as, for a moisture content lower than 0.1g starting from DWS values, using the relationship:
The DWS values are linearly related to the average value of the differential hydration enthalpy for tissue moisture contents lower than 0.10 g H2O/ g dry weight. To obtain estimates of the average hydration enthalpies of materials different from durum wheat leaves, calibration regression lines should be experimentally constructed: DWS values versus average differential hydration enthalpy. For this, it is necessary: (a) to draw several isotherm curves of samples after equilibrating them at several relative humidities (usually 15-20, or more) and at two temperatures; and (b) to calculate the thermodynamic parameters according to the Clausius-Clapeyron equation for heat of vaporisation at different tissue water contents (Rascio et al. 1997).
LINKS TO RESOURCES AND SUPPLIERS
https://www.academia.edu/7165913/ASSIGNMENT_1_WATER_ACTIVITY
LITERATURE REFERENCES
Wolf W, Spiess WE, Jung G (1985) Standarization of isotherm measurement (COST-project 90 and 90 bis). In D. Simatos, & J. L. Multon (Eds.), Properties of water in foods. Dordrecht, The Netherlands: Martinus Nijhoff Publishers.(pp. 661-679).
Rascio A, Russo M, Di Fonzo N (1997) Simplified procedure for estimating tissue affinity for bound water in durum wheat. Crop Science 37, 275-277.
Vertucci CW, Leopold AC (1987) The Relationship between Water Binding and Desiccation Tolerance in tissues. Plant Physiology 85, 232-238.
HEALTH, SAFETY & HAZARDOUS WASTE DISPOSAL CONSIDERATIONS
Chromic acid – sulphuric acid mixture should be prepared inside the chemical fume hoods, because is toxic and carcinogenic. To clean the weighting bottles you can substitute such cleaning solutions, with ultrasonic baths or biodegradable surfactants.